Open Mitral Valve Repair
Mitral repair does not require replacing the valve and keeps the majority of the native valve intact. Many studies have shown that there is a survival benefit with mitral repair vs. mitral replacement. Mitral repair has shown 1-2% mortality where replacement is 10.7% mortality. Mitral repair is less invasive than replacement as the incision for repair is smaller. Surgical mitral valve repair involves reshaping and removing portions of the leaflets in conjunction with annuloplasty, shrinking the diameter of the annulus. The benefit to this procedure is that the majority of the patient’s tissue is still intact and the dynamics of the left ventricle remain unchanged.
Despite the fact that valve repair has proven to be more effective than valve replacement, there are still many drawbacks. Repair techniques are more complicated than replacement and require greater skill of the surgeon to complete. Repair operation also takes longer than replacement. An average mitral repair takes 3-6 hours where an average mitral replacement takes 1-2 hours. This means the patient is on cardiac bypass for a longer amount of time, which increases the risks of side effects.
Despite the fact that valve repair has proven to be more effective than valve replacement, there are still many drawbacks. Repair techniques are more complicated than replacement and require greater skill of the surgeon to complete. Repair operation also takes longer than replacement. An average mitral repair takes 3-6 hours where an average mitral replacement takes 1-2 hours. This means the patient is on cardiac bypass for a longer amount of time, which increases the risks of side effects.
In Depth
Currently the most common technique for mitral regurgitation is quadrangle resection of the posterior leaflet with annuloplasty. For this technique, if the leaflet is greater than 1.5 cm, a procedure invented by Carpentier is used called sliding leaflet plasty of the posterior leaflet. Figure 4 shows the sutures involved in Carpentier’s technique. (7) This involves changing an extensive portion of the posterior annulus. (16) Sliding repair is designed to reduce the risk of systolic anterior motion (SAM). (7) Carpentier’s procedure has been implemented since 1970 and has long-term satisfactory results. This procedure requires no need for long-term anticoagulation and shows no signs for risk of thromboembolism. (5) For this procedure a 10-year freedom of reoperation was 93%, however anterior leaflet prolapse showed less successful results. In optimal cases that used quadrangle posterior resection with annuloplasty and confirmation by echocardiography, the 10-year freedom of reoperation was 98%. (7) However, one risk factor showed excessive tension on posterior leaflet during attachment after quadrangle resection. (16)
For anterior leaflet prolapse, which is less common than posterior leaflet prolapse, different techniques are used. Due to the greater complications associated with anterior leaflet prolapse, physicians are much less inclined to operate on the anterior leaflet than the posterior leaflet to repair mitral regurgitation. (5) There are greater complications associated with anterior leaflet repair because it is a larger leaflet so trying to resect a portion of the anterior leaflet requires a more complicated procedure. The anterior leaflet is also more difficult to reconstruct surgically.
The most common repair techniques for the anterior leaflet are transferring chords from posterior leaflet or adjacent areas of the anterior leaflet prolapse to the region of the anterior leaflet that needs a chordae tendineae, creation of artificial chords, and Alfieri edge-to-edge repair. Annuloplasty is used in in conjunction with these techniques. (7)
Creation of artificial chords with expanded polytetrafluoroethylene (e-PTFE)has become increasingly popular because of its accessibilityand long-term durability. However, challenges to this procedure include determining correct chordal length and tying the knot without slipping. In a 20-year study of 608 patients who underwent artificial chordae creation with e-PTFE, calcification of GORTEX was never reported. (18) Risk of operation increased with anterior leaflet prolapse, chordal shortening, lack of intraoperative echocardiography, and failure to use an annuloplasty. (7)
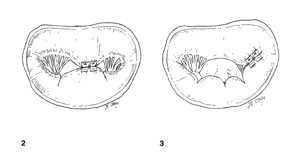
The double orifice mitral valve, shown in Figure 5.2 and 5.3 is created by estimating the free edges of the mitral valve and suturing together a section of the posterior and anterior leaflet. It also usually requires an annuloplasty ring. Operative deaths occurring with in 30 days of operation occurred in 1.3% of patients, reoperation occurred in 8% of patients. (5)
The above techniques showed improved results when annuloplasty was used in conjunction with the operation. However, a lab study in a normal beating ovine heart published in September 2011 showed that annuloplasty rings alter native annular dimensions, dynamics, and shape. This resulted in changing anterior mitral leaflet strain patterns, impaired flow from atrium to ventricle, and shortened long-term repair durability. (3)
Currently the majority of mitral valve operations are performed using bicaval venous cannulation, mild systemic hypothermia, and intermittent cold-blood cardioplegia. The procedures are performed on heart-lung bypass with the surgeon entering the heart through the left atrium, which allows the physician a clear view of the mitral valve. (13) Most of these procedures require annuloplasty, resection of the prolapsed leaflet, and/or artificial chordae replacement. (6)
There are complications that are associated with putting a patient on cardiac bypass. These include Postperfusion syndrome (Pumphead), hemolysis, capillary leak syndrome, and clotting of blood in the circuit or air embolism. During cardiac bypass cauterizing device is used to cut and seal soft tissue and to burn edges shut. Bits from the cauterizing mix with the blood during the procedure and suction is used every 10 to 20 minutes for 10-15 seconds. This material is always passed through a filter before it is put back into the circulating blood system. The problem of filtering the blood is exposing the blood to a large surface are of a manmade surface. It has also been shown that neural behavioral problems are common after cardiac bypass. It is believed to be due to reduced cerebral blood flow due to microemboli. The microemboli are believed to be due to the oxygenator in the heart-lung machine. (13)
Many patients forgo mitral valve surgery due to the many side effects and the 1% mortality rate risk. Our minimally invasive catheter based off-pump procedure would allow for these patients to undergo mitral regurgitation repair without the risk factors from going on cardiac bypass. This would increase patient use, reduce recovering time, and reduce cost of the procedure due to less equipment and less physicians needed in the operating room during the procedure. (13)
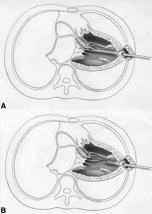
Recently a technique has been used for mitral valve repair by artificial chordae replacement. It is performed in a minimally invasive, closed chest off-pump procedure. Performing the procedure while the heart is beating allows for the surgeon to adjust the length of the chord while looking at real-time echocardiography as a guide to see what tension provides minimal regurgitation. A 3-4 inch incision is made in order to access the heart by left lateral thoracotomy. Using transesophageal echocardiography as guidance, the NEOCHORD device punctures the apex of the left ventricle. The NEOCHORD device then adheres to the prolapsed mitral leaflet and secures an artificial chordae tendineae (5-0 Gore-Tex) to its edge. This is shown in Figure 6A & B. After the surgeon is satisfied with the MR reduction, the suture is tied and cut. The 3-4 inch incision is sutured and the surgery is complete. The NEOCHORD device is currently under clinical investigation and is not yet approved for sale. (2)
An in vitro study showed that the technique of implanting an artificial chordae tendineae from the mitral valve leaflet to the apex of the left ventricular wall while the heart is still beating generates tension vectors that are in different direction than the tension of the physiological chordae of the heart. With increased time this could lead to early failure of the mitral valve prolapse repair. However, results also showed that the new ACTs were in the same ranges of tension as the native chordae. Using a transapical approach by puncturing the apex also could lead to complications due to the bulkiness of the device. A transaortic procedure would be ideal as it only has one small entry point through the femoral artery and also does not require a 3-4 inch incision for a left lateral thoracotomy. (2)
For anterior leaflet prolapse, which is less common than posterior leaflet prolapse, different techniques are used. Due to the greater complications associated with anterior leaflet prolapse, physicians are much less inclined to operate on the anterior leaflet than the posterior leaflet to repair mitral regurgitation. (5) There are greater complications associated with anterior leaflet repair because it is a larger leaflet so trying to resect a portion of the anterior leaflet requires a more complicated procedure. The anterior leaflet is also more difficult to reconstruct surgically.
The most common repair techniques for the anterior leaflet are transferring chords from posterior leaflet or adjacent areas of the anterior leaflet prolapse to the region of the anterior leaflet that needs a chordae tendineae, creation of artificial chords, and Alfieri edge-to-edge repair. Annuloplasty is used in in conjunction with these techniques. (7)
Creation of artificial chords with expanded polytetrafluoroethylene (e-PTFE)has become increasingly popular because of its accessibilityand long-term durability. However, challenges to this procedure include determining correct chordal length and tying the knot without slipping. In a 20-year study of 608 patients who underwent artificial chordae creation with e-PTFE, calcification of GORTEX was never reported. (18) Risk of operation increased with anterior leaflet prolapse, chordal shortening, lack of intraoperative echocardiography, and failure to use an annuloplasty. (7)
The double orifice mitral valve, shown in Figure 5.2 and 5.3 is created by estimating the free edges of the mitral valve and suturing together a section of the posterior and anterior leaflet. It also usually requires an annuloplasty ring. Operative deaths occurring with in 30 days of operation occurred in 1.3% of patients, reoperation occurred in 8% of patients. (5)
The above techniques showed improved results when annuloplasty was used in conjunction with the operation. However, a lab study in a normal beating ovine heart published in September 2011 showed that annuloplasty rings alter native annular dimensions, dynamics, and shape. This resulted in changing anterior mitral leaflet strain patterns, impaired flow from atrium to ventricle, and shortened long-term repair durability. (3)
Currently the majority of mitral valve operations are performed using bicaval venous cannulation, mild systemic hypothermia, and intermittent cold-blood cardioplegia. The procedures are performed on heart-lung bypass with the surgeon entering the heart through the left atrium, which allows the physician a clear view of the mitral valve. (13) Most of these procedures require annuloplasty, resection of the prolapsed leaflet, and/or artificial chordae replacement. (6)
There are complications that are associated with putting a patient on cardiac bypass. These include Postperfusion syndrome (Pumphead), hemolysis, capillary leak syndrome, and clotting of blood in the circuit or air embolism. During cardiac bypass cauterizing device is used to cut and seal soft tissue and to burn edges shut. Bits from the cauterizing mix with the blood during the procedure and suction is used every 10 to 20 minutes for 10-15 seconds. This material is always passed through a filter before it is put back into the circulating blood system. The problem of filtering the blood is exposing the blood to a large surface are of a manmade surface. It has also been shown that neural behavioral problems are common after cardiac bypass. It is believed to be due to reduced cerebral blood flow due to microemboli. The microemboli are believed to be due to the oxygenator in the heart-lung machine. (13)
Many patients forgo mitral valve surgery due to the many side effects and the 1% mortality rate risk. Our minimally invasive catheter based off-pump procedure would allow for these patients to undergo mitral regurgitation repair without the risk factors from going on cardiac bypass. This would increase patient use, reduce recovering time, and reduce cost of the procedure due to less equipment and less physicians needed in the operating room during the procedure. (13)
Recently a technique has been used for mitral valve repair by artificial chordae replacement. It is performed in a minimally invasive, closed chest off-pump procedure. Performing the procedure while the heart is beating allows for the surgeon to adjust the length of the chord while looking at real-time echocardiography as a guide to see what tension provides minimal regurgitation. A 3-4 inch incision is made in order to access the heart by left lateral thoracotomy. Using transesophageal echocardiography as guidance, the NEOCHORD device punctures the apex of the left ventricle. The NEOCHORD device then adheres to the prolapsed mitral leaflet and secures an artificial chordae tendineae (5-0 Gore-Tex) to its edge. This is shown in Figure 6A & B. After the surgeon is satisfied with the MR reduction, the suture is tied and cut. The 3-4 inch incision is sutured and the surgery is complete. The NEOCHORD device is currently under clinical investigation and is not yet approved for sale. (2)
An in vitro study showed that the technique of implanting an artificial chordae tendineae from the mitral valve leaflet to the apex of the left ventricular wall while the heart is still beating generates tension vectors that are in different direction than the tension of the physiological chordae of the heart. With increased time this could lead to early failure of the mitral valve prolapse repair. However, results also showed that the new ACTs were in the same ranges of tension as the native chordae. Using a transapical approach by puncturing the apex also could lead to complications due to the bulkiness of the device. A transaortic procedure would be ideal as it only has one small entry point through the femoral artery and also does not require a 3-4 inch incision for a left lateral thoracotomy. (2)
Carpentier’s technique of valvular reconstruction.
Figure A: Instrument inserted into left ventricle grabbing onto free edge of prolapse leaflet and securing 5-0 Gore-Tex to its free edge.
Figure B: Instrument is removed from left ventricle and artificial chordae is secured outside of the left ventricular apex.
Figure B: Instrument is removed from left ventricle and artificial chordae is secured outside of the left ventricular apex.
Figure 2: “Edge-to-edge” technique to correct a prolapse of the middle portion of the anterior leaflet. This creates a double mitral orifice.
Figure 3: “Edge-to-edge” which corrects a prolapse of the anterior leaflet of the mitral valve in the proximity of the postero-medial commissure. This reduces the mitral orifice.
Figure 3: “Edge-to-edge” which corrects a prolapse of the anterior leaflet of the mitral valve in the proximity of the postero-medial commissure. This reduces the mitral orifice.