Percutaneous Mitral Valve Repair
Percutaneous mitral valve repair is a catheter-based approach that reduces the side effects associated with open mitral repair and replacement. Percutaneous repair is done off-pump and therefore does not have the complications associated with cardioplegia and also maintains the physiology of the heart which is believed to increase life-expectancy and coaptation of the mitral valve. Percutaneous mitral valve repair techniques are still being developed. From a design standpoint, there are many limitations in repairing the mitral valve percutaneously – one key limitation is the size of the device. One major benefit of percutaneous repair is that patients who forwent open mitral vale repair due to the risk of cardiac bypass will now be able to undergo the repair. Percutaneous treatment is associated with increased safety, faster recovery time, cheaper procedures, improved left ventricular dimensions, and clinical improvements in quality of life. Long-term follow-up is intended to provide additional data to better understand the risks and benefits to percutaneous treatment of mitral regurgitation. (4)
In Depth
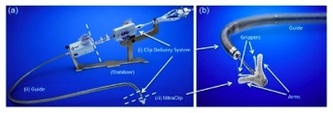
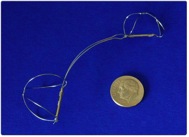
A percutaneous device for mitral regurgitation has been developed that requires a mechanical MitraClip, which grabs onto the free-edges of the leaflets and clamps down creating a double-orifice. The MitraClip is delivered into the left atrium through transseptal catheterization. The MitraClip device is a 4-mm-wide cobalt–chromium implant consisting of two arms that open and close with the clip delivery system handle. The system consists of two principal components, shown in Figure &A & 7B:
· Clip delivery system (CDS) used to position and implement the clip
· Steerable guide catheter
The MitraClip device is attached to the CDS. A stabilizer keeps the system in a precise position. In a 107-patient study, the MitraClip repair significantly reduced mitral regurgitation. (4)
Figure 7 A: Clip delivery System Figure 7 B: MitraClip device
The patient is under general anesthesia and the MitraClip is guided with the use of fluoroscopic and transesophageal echocardiographic to reach the inferior vena cava. Atrial transseptal puncture is completed, which allows the device to be steered until it is aligned over the mitral valve regurgitation area. The device grabs onto the free leaflets and the MitraClip clamps down on the anterior and posterior leaflets. Echocardiography is used to assess a grade of 2+ or less mitral regurgitation. Patients were treated with heparin during the procedure, with aspirin for 6 months and with clopidogrel for 30 days after the procedure. (4)
Percutaneous valve repair reduced the risk of any major adverse event within in 30 days of the operation as compared to open valvular repair surgery. However, MitraClip percutaneous repair was less effective at reducing mitral regurgitation than open surgery before hospital discharge. The rates of mitral regurgitation reduction evened out at 12 and 24 months. Another potential drawback to the MitraClip device is that the device is so big it has to puncture the septum of the heart, which can lead to further complications. The procedure also does not maintain the physiology of the heart and creates a double-orifice in the valve, which is not the native structure of the mitral valve. (4)
The CARILLON device procedure is another percutaneous mitral valve repair, except it manipulates the coronary sinus to constrict the mitral valve instead of dealing with the mitral valve directly. During the CARILLON device procedure the patient is put under general anesthesia and a transesophageal echocardiography is used to guide the 9F catheter. The coronary sinus is cannulated and the arteriovenous anatomy’s dimensions are taken in order to implement an appropriately sized CARILLON device. The CARILLON device, shown in Figure 8, is a fixed length implant with a double-anchor made of nitinol. The delivery catheter is guided through the superior vena cava to the coronary sinus and then the CARILLON device is threaded through the catheter. Once the device is in the correct placement of the coronary sinus near the anterior commissure of the mitral valve, the delivery catheter is retracted to enable passive expansion of the CARILLON device. The delivery catheter then pushes the device so that the device expands to its full potential. The anchors of the device are oversized compared to the diameter of the coronary artery which creates pressure on the inside walls of the coronary sinus. The expansion of the coronary sinus puts pressure on the mitral valve. This causes increased coaptation of the mitral valve leaflets, which reduces mitral regurgitation. (19)
Drawbacks to this device implantation included a 43% removal of the device, the major causes being insufficient mitral regurgitation reduction and coronary artery compromise. The anatomy the coronary sinus varies from patient to patient is not always complete and consistently near the mitral annulus. These cases would lead the CARILLON device obsolete. There was a 2.2% death rate 30 days or earlier to the operation. Further studies need to be conducted in order to define the long-term efficacy of the study. (19)
· Clip delivery system (CDS) used to position and implement the clip
· Steerable guide catheter
The MitraClip device is attached to the CDS. A stabilizer keeps the system in a precise position. In a 107-patient study, the MitraClip repair significantly reduced mitral regurgitation. (4)
Figure 7 A: Clip delivery System Figure 7 B: MitraClip device
The patient is under general anesthesia and the MitraClip is guided with the use of fluoroscopic and transesophageal echocardiographic to reach the inferior vena cava. Atrial transseptal puncture is completed, which allows the device to be steered until it is aligned over the mitral valve regurgitation area. The device grabs onto the free leaflets and the MitraClip clamps down on the anterior and posterior leaflets. Echocardiography is used to assess a grade of 2+ or less mitral regurgitation. Patients were treated with heparin during the procedure, with aspirin for 6 months and with clopidogrel for 30 days after the procedure. (4)
Percutaneous valve repair reduced the risk of any major adverse event within in 30 days of the operation as compared to open valvular repair surgery. However, MitraClip percutaneous repair was less effective at reducing mitral regurgitation than open surgery before hospital discharge. The rates of mitral regurgitation reduction evened out at 12 and 24 months. Another potential drawback to the MitraClip device is that the device is so big it has to puncture the septum of the heart, which can lead to further complications. The procedure also does not maintain the physiology of the heart and creates a double-orifice in the valve, which is not the native structure of the mitral valve. (4)
The CARILLON device procedure is another percutaneous mitral valve repair, except it manipulates the coronary sinus to constrict the mitral valve instead of dealing with the mitral valve directly. During the CARILLON device procedure the patient is put under general anesthesia and a transesophageal echocardiography is used to guide the 9F catheter. The coronary sinus is cannulated and the arteriovenous anatomy’s dimensions are taken in order to implement an appropriately sized CARILLON device. The CARILLON device, shown in Figure 8, is a fixed length implant with a double-anchor made of nitinol. The delivery catheter is guided through the superior vena cava to the coronary sinus and then the CARILLON device is threaded through the catheter. Once the device is in the correct placement of the coronary sinus near the anterior commissure of the mitral valve, the delivery catheter is retracted to enable passive expansion of the CARILLON device. The delivery catheter then pushes the device so that the device expands to its full potential. The anchors of the device are oversized compared to the diameter of the coronary artery which creates pressure on the inside walls of the coronary sinus. The expansion of the coronary sinus puts pressure on the mitral valve. This causes increased coaptation of the mitral valve leaflets, which reduces mitral regurgitation. (19)
Drawbacks to this device implantation included a 43% removal of the device, the major causes being insufficient mitral regurgitation reduction and coronary artery compromise. The anatomy the coronary sinus varies from patient to patient is not always complete and consistently near the mitral annulus. These cases would lead the CARILLON device obsolete. There was a 2.2% death rate 30 days or earlier to the operation. Further studies need to be conducted in order to define the long-term efficacy of the study. (19)
Figure 7 A: Clip delivery System Figure 7 B: MitraClip device
Figure 8: The CARILLON Mitral Contour System is a fixed length, double-anchor, nitinol device designed to be positioned within the coronary sinus to reduce Type II mitral regurgitation.